premiumtimesng.com
NAFDAC restates ban on multi - dose anti - malaria suspension
premiumtimesng.com · Feb 27, 2026 · Collected from GDELT
Summary
Published: 20260227T191500Z
Full Article
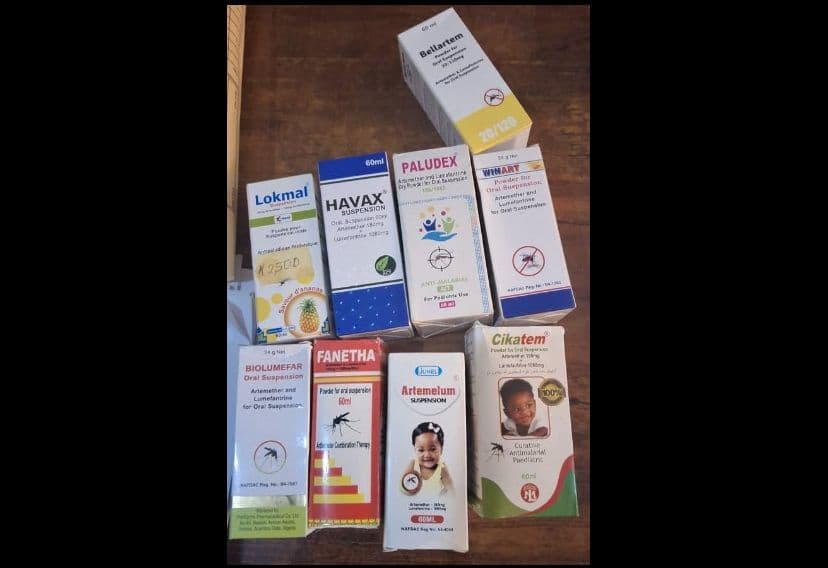
The National Agency for Food and Drug Administration and Control (NAFDAC) has restated its regulatory directive discontinuing the registration of Anti-Malaria multi-dose, artemether/lumefantrine dry powder for oral suspension, warning that the formulation becomes unstable after reconstitution and may lose efficacy over time. In a Public Alert issued on Friday, the agency reminded stakeholders that the directive, first issued in March last year, remains in force and applies to all locally manufactured and imported multi-dose artemether/lumefantrine dry powder for oral use. NAFDAC had, in an earlier advisory announced the discontinued registration of the product following findings from stability studies. At the time, the agency said it would no longer accept new applications, renewal applications or variation applications for any local or imported multi-dose artemether/lumefantrine dry powder for oral suspension. The suspension covered all brands and manufacturers of the product. Risk statement According to NAFDAC, stability studies demonstrated that reconstituted artemether/lumefantrine oral suspension becomes unstable after mixing, resulting in a loss of efficacy. The agency warned that when a medicine loses its effectiveness, it may lead to worsening of the condition being treated, increased risk of complications, delays in appropriate treatment and, in severe cases, death. In the renewed advisory, NAFDAC stressed that reconstituted anti-malarial suspensions are unstable and may no longer deliver the required therapeutic effect. Formulation switch Reaffirming its earlier position, the agency said it no longer accepts new, renewal or variation applications for the affected product. It directed manufacturers who wish to continue producing artemether/lumefantrine formulations to adopt safer and more stable alternatives, including dispersible tablets or powders or granules packaged in sachets for single-dose use. According to NAFDAC, single-dose formulations help prevent the instability issues associated with multi-dose suspensions after reconstitution. READ ALSO: Sachet Alcohol Ban: Health ministry says it lacks power to stop NAFDAC enforcement Surveillance, public reporting The directive applies to all brands of multi-dose artemether/lumefantrine dry powder for oral suspension and to all manufacturers and importers. Healthcare professionals and consumers have been advised to report any suspected sale of the discontinued products, as well as substandard or falsified medicines and medical devices, to the nearest NAFDAC office. Reports can also be made through the agency’s toll-free number, 0800-162-3322, or via email at [email protected].
Share this story